
Research & Publications
Selected and Representative Abstracts from Publications and Meetings
Sidestream Smoke Affects Dendritic Complexity and Astrocytes After Model Mild Closed Head Traumatic Brain Injury
- Cell Mol Neurobiol . 2022 Jul;42(5):1453-1463.
- doi: 10.1007/s10571-020-01036-5. Epub 2021 Jan 8
Abstract
Effect of Mild Blast-Induced TBI on Dendritic Architecture of the Cortex and Hippocampus in the Mouse
- Scientific Reports. 2020 Feb 10;10(1):2206
- doi: 10.1038/s41598-020-59252-4.
Abstract
Traumatic brain injury (TBI) has been designated as a signature injury of modern military conflicts. Blast trauma, in particular, has come to make up a significant portion of the TBIs which are sustained in warzones. Though most TBIs are mild, even mild TBI can induce long term effects, including cognitive and memory deficits. In our study, we utilized a mouse model of mild blast-related TBI (bTBI) to investigate TBI-induced changes within the cortex and hippocampus. We performed rapid Golgi staining on the layer IV and V pyramidal neurons of the parietal cortex and the CA1 basilar tree of the hippocampus and quantified dendritic branching and distribution. We found decreased dendritic branching within both the cortex and hippocampus in injured mice. Within parietal cortex, this decreased branching was most evident within the middle region, while outer and inner regions resembled that of control mice. This study provides important knowledge in the study of how the shockwave associated with a blast explosion impacts different brain regions.
Mild Blast-Related TBI in a Mouse Model Alters Amygdalar Neurostructure and Circuitry
- Exp Neurol. 2019 May;315:9-14
- doi: 10.1016/j.expneurol.2019.01.020. Epub 2019 Jan 31.
Abstract
Background: Traumatic brain injury (TBI) is a widespread public health problem and a signature injury of our military in modern conflicts. Despite the long-term effects of even mild brain injuries, an effective treatment remains elusive. Coffee and several of its compounds, including caffeine, have been identified as having neuroprotective effects in studies of neurodegenerative disease. Given the molecular similarities between TBI and neurodegenerative disease, we have devised a study to test a nanocoffee extract in the treatment of a mouse model of mild TBI.
Results: After a single injury and two subsequent injections of nanocoffee, we identified treatment as being associated with improved behavioral outcomes, favorable molecular signaling changes, and dendritic changes suggestive of improved neuronal health.
Conclusions: We have identified coffee extracts as a potential viable multifaceted treatment approach to target the secondary injury associated with TBI.
Behavior, Protein, and Dendritic Changes After Model Traumatic Brain Injury and Treatment With Nanocoffee Particles
- BMC Neurosci . 2019 Aug 22;20(1):44.
- doi: 10.1186/s12868-019-0525-5.
Abstract
Traumatic brain injury (TBI) continues to be a signature injury of our modern conflicts. Due in part to increased use of improvised explosive devices (IEDs), we have seen blast trauma make up a significant portion of TBIs sustained by deployed troops and civilians. In addition to the physical injury, TBI is also a common comorbidity with post-traumatic stress disorder (PTSD). Previous research suggests that PTSD is often associated with increased signaling within the amygdala, leading to feelings of fear and hyperarousal. In our study, we utilized a mouse model of mild blast-related TBI (bTBI) to investigate how TBI induces changes within the amygdala, which may provide favorable conditions for the development of PTSD. To do this, we performed Golgi staining on the stellate neurons of the basolateral amygdala and quantified dendritic amount, distribution, and complexity. We found increases in dendritic branching and in the density of dendritic spines in injured mice. Increases in spine density appears to be primarily due to increases in memory associated mushroom type dendritic spines. These changes observed in our bTBI model that are consistent with chronic stress models, suggesting an important connection between the physical changes induced by TBI and the neurological symptoms of PTSD.
Repetitive Mild Closed Head Injury Alters Protein Expression and Dendritic Complexity in a Mouse Model
- J Neurotrauma. 2018 Jan 1;35(1):139-148
- doi: 10.1089/neu.2017.5070. Epub 2017 Aug 23.
Jessica N Saykally 1 2, Whitney A Ratliff 1 2, Kristen L Keeley 1 2, Chaim G Pick 3, Ronald F Mervis 4 5, Bruce A Citron 1 2
Abstract
Worldwide head injuries are a growing problem. In the United States alone, 1.7 million people suffer a head injury each year. While most of these injuries are mild, head injury sufferers still sustain symptoms that can have major medical and economical impacts. Moreover, repetitive mild head injuries, like those observed in active military personnel and athletes, have demonstrated a more severe and long-term set of consequences. In an effort to better understand the delayed pathological changes following multiple mild head injuries, we used a mouse model of mild closed head injury (with no motor deficits observed by rotarod testing) and measured dendritic complexity at 30 days after injury and potentially related factors up to 60 days post-injury. We found an increase in TDP-43 protein at 60 days post-injury in the hippocampus and a decrease in autophagy factors three days post-injury. Alterations in dendritic complexity were neuronal subtype and location specific. Measurements of neurotropic factors suggest that an increase in complexity in the cortex may be a consequence of neuronal loss of the less connected neurons.
Rescue of the Functional Alterations of Motor Cortical Circuits in Arginase Deficiency by Neonatal Gene Therapy
- J Neurosci. 2016 Jun 22;36(25):6680-90
- doi: 10.1523/JNEUROSCI.0897-16.2016.
Abstract
Arginase 1 deficiency is a urea cycle disorder associated with hyperargininemia, spastic diplegia, loss of ambulation, intellectual disability, and seizures. To gain insight on how loss of arginase expression affects the excitability and synaptic connectivity of the cortical neurons in the developing brain, we used anatomical, ultrastructural, and electrophysiological techniques to determine how single-copy and double-copy arginase deletion affects cortical circuits in mice. We find that the loss of arginase 1 expression results in decreased dendritic complexity, decreased excitatory and inhibitory synapse numbers, decreased intrinsic excitability, and altered synaptic transmission in layer 5 motor cortical neurons. Hepatic arginase 1 gene therapy using adeno-associated virus rescued nearly all these abnormalities when administered to neonatal homozygous knock-out animals. Therefore, gene therapeutic strategies can reverse physiological and anatomical markers of arginase 1 deficiency and therefore may be of therapeutic benefit for the neurological disabilities in this syndrome.
Significance statement: These studies are one of the few investigations to try to understand the underlying neurological dysfunction that occurs in urea cycle disorders and the only to examine arginase deficiency. We have demonstrated by multiple modalities that, in murine layer 5 cortical neurons, a gradation of abnormalities exists based on the functional copy number of arginase: intrinsic excitability is altered, there is decreased density in asymmetrical and perisomatic synapses, and analysis of the dendritic complexity is lowest in the homozygous knock-out. With neonatal administration of adeno-associated virus expressing arginase, there is near-total recovery of the abnormalities in neurons and cortical circuits, supporting the concept that neonatal gene therapy may prevent the functional abnormalities that occur in arginase deficiency.
Neonatal infection with Borna Disease Virus causes widespread dendritic branching and spine neuropathology in brains of 3 week-old rats
- Presented at the Society for Neuroscience, San Diego (2016)
Abstract
There is much concern surrounding the neuropathological sequelae of gestational Zika virus infection in man. Here, we examined the effects of another neurotropic RNA virus, the Bornavirus, on the developing brain of rat pups. Information regarding the impact of this viral infection on developing neurons and neural circuitry would lend additional insight (and could raise red flags) regarding the potential neurodevelopmental consequences of other related fetal or neonatal viral infections. Neonatal Lewis rats were infected with Borna Disease Virus (BDV) by innoculation into the right cerebral hemisphere (N=6). Controls (N=6) received PBS. Three week-old rats were sacrificed and brains stained using Rapid Golgi (hemispheres) or Golgi-Cox (cerebellum) procedures. Dendritic branching and spines were quantitated from coded slides. BDV-infected rat brains tended to show widespread dysmorphic changes in neurons characterized by reduced branching of the dendritic arbor and loss of complexity of the tree, and of dendritic spine loss, often accompanied by dendritic branch varicosities Significant effects of the viral infection were seen in all regions evaluated. The most salient reductions were found were found in: (1) Cerebellar Purkinje cells: branching area (-21%), spines (-16%); (2) Hippocampal CA1s: basilar branching (-20%), spines (-9%); (3) Dentate Gyrus Granule cells: branching (-29%), spines (-25%, thin spines); (4) Parietal Cortex Layer V pyramids: basilar branching (-19%), spines (-12%). These findings suggest that early exposure to a neurotropic RNA virus can disrupt maturation of normal dendritic parameters, impair neuroplasticity, and damage brain circuitry. As such, these virus-related dysmorphic changes may represent an underlying neurostructural basis for the pathogenesis of subsequent cognitive impairment and/or developmental disorders.
Mild Blast-Related TBI in a Mouse Model Alters Amygdalar Neurostructure & Circuitry: Relevance to PTSD
- Presented at the annual meeting of the National Neurotrauma Society, Lexington, KY (June, 2016)
1 Neurostructural Research Labs, Neurotrauma Studies Group, Tampa, USA
2 The University of South Florida, The Honors College, Tampa, USA
3 Tel-Aviv University, Sackler School of Medicine, Anatomy and Anthropology, Tel-Aviv, Israel
4University of South Florida Morsani College of Medicine, Center for Aging and Brain Repair, Department of Neurosurgery, Tampa, FL
Blast-related traumatic brain injury (bTBI) from improvised explosive devices (IEDs) has been the signature injury of the war against terror for both combatants and civilians. TBI is regarded as a major risk factor for the development of Post-Traumatic Stress Disorder (PTSD). The amygdala plays a vital role in fear conditioning and emotion; human studies have shown that the amygdala demonstrates hyperactivity in PTSD patients. As part of a larger study characterizing neuronal damage in a mouse model of blast-related TBI (bTBI) we evaluated the neuropathological impact of a mild bTBI, e.g., the effects on dendritic branching and spines of stellate neurons from the basolateral amygdala. Anesthetized 7 week-old mice were exposed to a single explosion which generated a pressure wave of 5.5 psi (lbs/sq in) and sacrificed 72 hrs later. Using the Golgi impregnation technique, from coded slides, the dendritic branching and spines of basolateral amydgalar neurons were quantified (N=6 subjects/group, n=5 neurons/subject). The Sholl analysis was used to evaluate the extent and distribution of the dendritic arbor. Dendritic spines were evaluated in terms of total spine density and spine configurations.
Results: Although there were no significant differences in the amount or complexity of the dendritic tree as a result of the bTBI, there was a significantly higher spine density (+10%, p=0.03, unpaired T test) on the amydgalar neurons in the bTBI group. Moreover, this increase was most pronounced for M-type (mushroom) spines (+27%, p = 0.002), and this spine type may be linked to a stronger emotional memory of the event, thus precipitating the PTSD syndrome.
The Take-Home Message: Results suggest that the bTBI-induced increase in dendritic spines (especially M-type) on basolateral amydalar neurons may contribute to hyperactivity of this nucleus and may thus constitute the underlying neuroanatomcal basis for the PTSD sequelae.
Repetitive Concussions in the Mouse Affect Dendritic Branching of Various Neuronal Populations Differently: A Golgi Study
- Presented at the annual meeting of the National Neurotrauma Society, Lexington, KY (June, 2016)
1 Neurostructural Research Labs, Neurotrauma Studies Group, Tampa, USA
2 University of South Florida, College of Arts & Sciences, Tampa, USA
3 Bay Pines VA Healthcare System, Research & Development, Bay Pines, USA
4 University of South Florida Morsani College of Medicine, Molecular Medicine, Tampa, USA
5University of South Florida Morsani College of Medicine,Dept Neurosurgery, Ctr Aging & Brain Repair, Tampa, FL
Traumatic brain injury (TBI) from multiple concussions is strongly associated with contact sports. The relationship between repetitive concussions and neuronal damage is poorly understood. We assessed the effects of multiple concussions on dendritic branching of cortical, hippocampal, and striatal neurons in the adult mouse. Using a closed head injury paradigm, a 30 gram weight was dropped on the right temporal-parietal region of anesthetized mice (N = 3/group). There were 5 concussions, each with a 24 hour interval (5xTBI). After a recovery period of 30 days, the mice were sacrificed. Sham controls were handled identically. For coded slides, brains were stained using the Rapid Golgi method. For dendritic branching analysis, camera lucida drawings were prepared and analyzed for amount, distribution, and complexity of the dendritic arbor.
Results: Striatal neurons from the 5xTBI brains had significantly less dendritic arbor and complexity. For the hippocampal CA1s, there was no difference in the dendritic arbor between the two groups. Conversely, in granule cells of the dentate gyrus there was a significant increase in the amount of dendritic material in the 5xTBI mice. Layer V pyramids from the 5xTBI parietal cortex also showed a significant increase in dendritic branching. These disparate findings may be attributed to several factors: susceptibility or sensitivity to TBI-related damage; different recovery capabilities, and/or neuronal loss resulting in compensatory dendritic growth in surviving neurons.
The Bottom Line: Findings suggest that multiple concussions may cause a distortion of normal brain circuitry/information transfer which can result in compromised cognitive function.
The Dendrites of Granule Cell Layer Neurons Are the Primary Injury Sites in the "Brain Diabetes" Rat
- Behavioral Brain Research 2015 Mar 1;280:78-83
- doi: 10.1016/j.bbr.2014.11.041. Epub 2014 Dec 2
Abstract
We previously demonstrated that rats that receive dorsal third ventricle (3V) streptozotocin (STZ) injections (STZ-3V-rats) exhibit cognitive decline as measured by the Morris Water Maze (MWM) and can be used as an animal model of Alzheimer’s disease (AD). Immunohistochemical studies of the hippocampal formations of these animals have revealed significant changes in cerebral insulin signalling pathways, as well as marked increases of amyloid beta (Ab) deposition. Here, we performed Sholl analyses of granule cell layer dendrites and measured dendrite spine densities to assess the effect of STZ on hippocampal morphology. In STZ-3V rats as the results, more branching, complex dendrite arborisation, and increased soma size of the granule cells were observed, while spine densities were decreased in all three spine types. An intraventricular injection of a long-acting insulin analogue improved STZ-induced behavioural and immunohistochemical changes. Nevertheless, dendrite spine densities remained diminished, presumably due to overall null changes since new spine formation due to insulin stimulation has been compensated by loss of old spines. It is concluded that cognitive decline in the “Brain Diabetes” rats is primarily due to impaired intracerebral insulin signalling and the ultimate results were injured excitatory inputs through the perforant pathway.
Dendritic Loss in the Prefrontal Cortex of MPTP-Treated Monkeys
- Presented at the Society for Neuroscience (2014)
Abstract
MPTP (1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine) is a neurotoxin precursor to MPP+, which causes permanent symptoms of Parkinson’s disease by destroying dopaminergic neurons in the substantia nigra of the brain. Monkeys exposed to low doses of MPTP may show cognitive deficits without motor abnormalities. Such cognitive deficits are similar to that observed in early Parkinson’s Disease. The prefrontal cortex, which plays an important role in executive functions, receives a dense dopaminergic input. Dysregulation of dopamine (DA) circuitry may disrupt normal cognition. Using monkeys, the goal of this study was to assess the effects of MPTP neurotoxicity on dendritic circuitry in the monkey dorsolateral prefrontal cortex (DLPFC), a region critical to working memory performance. Young adult African Green monkeys were administered MPTP (2.25mg/kg) over 5 consecutive days. The animals were sacrificed 10 months after drug administration. Blocks of fixed brain tissue were Golgi impregnated and coded slides prepared. Randomly selected layer V and layer II-III pyramids were evaluated for amount of dendritic material and complexity of the dendritic arbors. Analysis of layer II-III pyramids showed a significant loss of arbor in the MPTP-treated monkeys (-22%) with a similar reduction in dendritic branching complexity. Layer V pyramids, by contrast, showed less disruption of circuitry with a 15% loss of dendritic branching and an 11% loss of neuronal complexity. These data support and extend previous findings on spine synapse changes following MPTP. The data is further consistent with a role for dopamine in regulating dendritic branching and spine changes on pyramidal neurons in the dorsolateral prefrontal cortex.
Repeated Administrations of Human Umbilical Cord Blood Cells Improve Disease Outcomes in a Mouse Model of Sanfilippo Syndrome Type III B
- Cell Transplant. 2014;23(12):1613-30.
- doi: 10.3727/096368914X676916.
Abstract
Sanfilippo syndrome type III B (MPS III B) is an inherited disorder characterized by a deficiency of α-N-acetylglucosaminidase (Naglu) enzyme leading to accumulation of heparan sulfate in lysosomes and severe neurological deficits. We have previously shown that a single administration of human umbilical cord mononuclear cells (hUCB MNCs) into Naglu knockout mice decreased behavioral abnormalities and tissue pathology. In this study, we tested whether repeated doses of hUCB MNCs would be more beneficial than a single dose of cells. Naglu mice at 3 months of age were randomly assigned to either a Media-only group or one of three hUCB MNC treatment groups–single low dose (3 × 10(6) cells), single high dose (1.8 × 10(7) cells), or multiple doses (3 × 10(6) cells monthly for 6 months) delivered intravenously; cyclosporine was injected intraperitoneally to immune suppress the mice for the duration of the study. An additional control group of wild-type mice was also used. We measured anxiety in an open field test and cognition in an active avoidance test prior to treatment and then at monthly intervals for 6 months. hUCB MNCs restored normal anxiety-like behavior in these mice (p < 0.001). The repeated cell administrations also restored hippocampal cytoarchitecture, protected the dendritic tree, decreased GM3 ganglioside accumulation, and decreased microglial activation, particularly in the hippocampus and cortex. These data suggest that the neuroprotective effect of hUCB MNCs can be enhanced by repeated cell administrations.
Potential neuroprotective benefits of coffee nano-particles in a mouse model of closed head traumatic brain injury: a morphometric analysis of dendritic parameters
- Presented at the Society for Neuroscience (2014)
Abstract
Millions of traumatic brain injuries (TBI) occur every year, 75% of which are considered mild traumatic brain injuries (mTBI). mTBIs are often seen in athletes, military personnel, and people involved in accidents. The cognitive effects of TBI are still untreatable. Neuronal loss and functional deficits are common attributes. Previous studies have suggested that coffee/caffeine can have neuroprotective, and even therapeutic, potential for brain diseases such as Alzheimer’s disease. Here, we investigated whether the beneficial effects of coffee may extend to treatments of mild TBI. There were 4 groups of mice: (1) vehicle + sham TBI; (2) vehicle + TBI; (3) coffee-treated + sham TBI; (4) coffee-treated + TBI. The coffee-treated mice were administered coffee nanoparticles (i.p.) at 30 minutes, 3 days, and 7 days following a mild TBI (a single 50 g weight drop-induced impact to the skull overlying the right temporal cortex). Brains were harvested 30 days following TBI. The Golgi impregnation method was used to visualize cortical layer V pyramids of the parietal cortex and the granule cells of the hippocampus for quantitative analysis of dendritic branching. Results showed that in the neocortex the dendritic domains of the layer V pyramids were significantly larger in both coffee groups (e.g., coffee + TBI and coffee + sham), suggesting either neuronal plasticity caused by the coffee nano-particles and/or compensatory dendritic hypertrophy associated with TBI-related neuronal death. Either – or both – events can occur independently of each other. In the hippocampus, both of the TBI groups had significantly less dendritic arbor than either of the sham TBI groups. This suggests TBI-related granule cell dendritic atrophy, but this cell population fails to exhibit any signs of neuroprotective properties in the coffee groups. Future stereology studies are planned to elucidate neuronal loss and compensatory mechanisms.
Developmental Neurotoxicity of 3,3',4,4'-tetrachloroazobenzene With Thyroxine Deficit: Sensitivity of Glia and Dentate Granule Neurons in the Absence of Behavioral Changes
- Toxics. 2014 Sep;2(3):496-532
- doi: 10.3390/toxics2030496.
Abstract
Thyroid hormones (TH) regulate biological processes implicated in neurodevelopmental disorders and can be altered with environmental exposures. Developmental exposure to the dioxin-like compound, 3,3′,4,4′-tetrachloroazobenzene (TCAB), induced a dose response deficit in serum T4 levels with no change in 3,5,3′- triiodothyronine or thyroid stimulating hormone. Female Sprague-Dawley rats were orally gavaged (corn oil, 0.1, 1.0, or 10 mg TCAB/kg/day) two weeks prior to cohabitation until post-partum day 3 and male offspring from post-natal day (PND)4-21. At PND21, the high dose showed a deficit in body weight gain. Conventional neuropathology detected no neuronal death, myelin disruption, or gliosis. Astrocytes displayed thinner and less complex processes at 1.0 and 10 mg/kg/day. At 10 mg/kg/day, microglia showed less complex processes, unbiased stereology detected fewer hippocampal CA1 pyramidal neurons and dentate granule neurons (GC) and Golgi staining of the cerebellum showed diminished Purkinje cell dendritic arbor. At PND150, normal maturation of GC number and Purkinje cell branching area was not observed in the 1.0 mg/kg/day dose group with a diminished number and branching suggestive of effects initiated during developmental exposure. No effects were observed on post-weaning behavioral assessments in control, 0.1 and 1.0mg/kg/day dose groups. The demonstrated sensitivity of hippocampal neurons and glial cells to TCAB and T4 deficit raises support for considering additional anatomical features of brain development in future DNT evaluations.
Four Weeks Lithium Treatment Alters Neuronal Dendrites in the Rat Hippocampus
- Int J Neuropsychopharmacol. 2013 Jul;16(6):1373-82
- doi: 10.1017/S1461145712001423. Epub 2013 Jan 18.
Abstract
A large body of evidence from molecular, cellular and human studies suggests that lithium may enhance synaptic plasticity, which may be associated with its therapeutic efficacy. However, only a small number of studies have directly assessed this. To determine whether lithium treatment alters structural synaptic plasticity, this study examined the effect of 4 wk lithium treatment on the amount and distribution of dendrites in the dentate gyrus (DG) and hippocampal area CA1 of young adult rats. Following 4 wk lithium or control chow feeding, animals were decapitated, the hippocampi were prepared and stained using a rapid Golgi staining technique and the amount and distribution of the dendritic branching was evaluated using Sholl analyses (method of concentric circles). In the DG, lithium treatment increased the amount and distribution of dendritic branches in the proximal half of dendritic trees of the granule cells and reduced branching in the distal half. In area CA1, the same treatment also increased the number of dendritic branches in the proximal half of apical dendritic trees of CA1 pyramidal cells and reduced branching in the distal half of apical dendritic trees but had no effect on basilar dendritic trees. The lithium treatment altered the total density of dendritic trees in neither the DG nor area CA1. These findings suggest that, in the DG and apical CA1, chronic lithium treatment rearranges neuronal morphology to increase dendritic branching and distribution to where major afferent input is received.
Effect of fractionated whole brain irradiation on neuronal morphology of rat cortex and hippocampus: Impact of late delayed brain injury
- Presented at the Radiation Research Society, New Orleans, September, 2013
Abstract
Fractionated partial or whole-brain irradiation (fWBI) is routinely used for the treatment of primary and metastatic brain cancer. Presently, ~200,000 patients/year are treated with fWBI, and ~ 100,000/year survive long enough (>6 months) to develop radiation-induced brain injury, including cognitive impairment, which can adversely impact their quality of life. Using a well-established fWBI rat model, the effects of fWBI on brain microcircuitry were evaluated 6 months following irradiation.
Methods: Six 10-12 week old F344xBN male rats were lightly anesthetized and received either: (1) 40 Gy fWBI or, (2) sham-irradiation. Coronal tissue blocks encompassing parietal cortex and hippocampus were Golgi stained. From coded slides, granule cells of the dentate gyrus (DG; 9 neurons/brain) and layer V pyramids (6 neurons/brain) were randomly selected for analysis of amount, distribution and complexity of the dendritic arbors. Dendritic spines were also evaluated on DG granule cells (5 neurons/brain).
Results: DG Granule Cell branching: Total branch length was not affected by fWBI however; the distribution of the arbor was shifted in the fWBI subjects with more dendritic material in the inner 2/3rds and less arbor in the outer 3rd of the tree. This was accompanied by a similar redistribution of dendritic complexity. Dendritic spines on the granule cells showed no significant change; however, Mushroom (M)-type spines in the neurons of fWBI subjects showed a 16% loss. Layer V Pyramidal Cell branching: A significant loss of dendritic arbor in the neurons of fWBI subjects was seen restricted to the inner half of the tree. This loss of arbor was mirrored by a reduction of dendritic arbor complexity.
Conclusions: Six months following 40 Gy fWBI, rats that received fWBI had significant alterations in dendritic morphology. Hippocampal and cortical neurons were significantly affected by distortion (granule cells) or reduction (layer V pyramids) of the arbor. Additionally, fWBI led to a significant decrease in the number of M-type spines in granular cells of the DG. These spines are thought to be related to memory function. In turn, these dendritic changes could negatively impact communication between neurons and represent a neurostructural basis for late-delayed radiation-induced cognitive impairment.
Neurotoxic effects of pre-weaning benzyl butyl phthalate (BBP) exposure on dendritic parameters in the rat
- Presented at the Society for Neuroscience, 2013
Abstract
Benzyl butyl phthalate (BBP) is a widely used industrial plasticizer which, if ingested, appears to have endocrine disrupting properties and may have deleterious consequences on the developing nervous system. In the present study, pregnant Sprague-Dawley rats were fed sweetened food pellets containing 10ug/ml of BBP. This dose was continued up until post-natal day 23 at which time the rat pups were sacrificed. Pregnant control rats received sweetened food pellets with no BBP. All brains were fixed in 10% neutral buffered formalin. Coronal blocks incorporating the parietal cortex and underlying hippocampus of 4 BBP-treated and 4 control 23 day-old male rats were Golgi stained. From coded slides camera lucida drawings of randomly selected granule cells of the dentate gyrus were prepared and evaluated for extent, complexity, and distribution of the dendritic arbor. Granule cell morphology was largely unaffected by this pre-weaning BBP exposure. Layer V pyramids, conversely, were significantly impacted by BBP. Analysis of the basilar dendritic tree showed that the BBP-exposed pups had significantly larger and more complex dendritic trees of the cortical pyramids. Dendritic spine density on the layer V pyramids was not significantly altered, but greater branching indicates that there was an overall increased synaptic presence in the layer V basilar tree. The present findings suggest that pre-weaning BBP exposure – e.g., during the period of the rodent brain growth spurt – may contribute to either additional cortical dendritic growth or diminished dendritic pruning in the course of the maturation of cortical circuits. Additional studies will determine if this is a transient or long-lasting phenomena, and if additional cell populations are also at risk from BBP exposure. Regardless, there is now evidence that environmental BBP may have deleterious consequences on neuronal development which could influence brain circuitry and, ultimately, behavior.
Dendritic Spine Alterations in the Hippocampus and Parietal Cortex of alpha7 Nicotinic Acetylcholine Receptor Knockout Mice
- Neuroscience. 2013 Mar 13;233:54-63
- doi: 10.1016/j.neuroscience.2012.12.025. Epub 2012 Dec 24.
1Boys Town National Research Hospital, Omaha, NE, USA.
2Neurostructural Research Labs, Tampa, FL
3Dept of Neurosurgery, Ctr of Excellence for Aging and Brain Repair, University of South Florida College of Medicine, Tampa, FL
Abstract
The α7 nicotinic acetylcholine receptor (nAChR) is involved in higher cognitive and memory functions, and is associated with the etiology of neurological diseases involving cognitive decline, including Alzheimer’s disease (AD). We hypothesized that spine changes in the α7 knockout might help to explain the behavioral deficits observed in α7 knockout mice and prodromal hippocampal changes in AD. We quantified several measures of dendritic morphology in the CA1 region of the mouse hippocampus in Golgi-stained material from wildtype and α7 knockout mice at P24. The most significant difference was a 64% increase in thin (L-type) dendritic spines on the CA1 basilar tree in knockout mice (p<.05). There were small decreases in the number of in N-type (-15%), M-type (-14%) and D-type (-4%) spine densities. The CA1 basilar dendritic tree of knockout mice had significantly less branching in the regions near the soma in comparison with wildtype animals (p<.01), but not in the more distal branching. Changes in the configuration of CA1 basilar dendritic spines have been observed in a number of experimental paradigms, suggesting that basilar dendritic spines are highly plastic. One component of cognitive dysfunction may be through α7-modulated GABAergic interneurons synapsing on CA1 basal dendrites.
Dendritic Alterations in the transthyretin (TTR) knockout mouse
Presented at the meeting of the Society for Neuroscience, New Orleans (2012)
BUCKINGAM3, M. ARADI3, P. SAWANGKUM3, R. CROUSE3, N. ARRUDA3.,J. N. BUXBAUM4
1Neurosurg. & Brain Repair, Univ. South Florida Coll Med., TAMPA, FL; 2Neurostructural Res. Labs, Inc., Tampa, FL; 3The Honors College,
Univ.of South Florida, Tampa, FL; 4Mol. & Exptl. Med., The Scripps Res. Inst., La Jolla, CA
Abstract
Dendritic Morphology of Amygdala and Hippocampal Neurons in More and Less Predator Stress Responsive Rats and More and Less Spontaneously Anxious Handled Controls
- Behav Brain Res. 2012 Jan 1; 226(1): 133–146.
- doi: 10.1016/j.bbr.2011.09.009
Robert Adamec,a,* Mark Hebert,a Jacqueline Blundell,a and Ronald F. Mervisb,
bCenter of Excellence for Aging and Brain Repair, Department of Neurosurgery and Brain Repair, University of South Florida College of Medicine, Tampa, FL 33612, USA cNeurostructural Research Labs, Inc., 12409 Telecom Drive, Tampa, FL 33637, USA
Abstract
We investigated the neurobiological bases of variation in response to predator stress (PS). Sixteen days after treatment (PS or handling), rats were grouped according to anxiety in the elevated plus maze (EPM). Acoustic startle was also measured. We examined the structure of dendritic trees of basolateral amygdala (BLA) output neurons (stellate and pyramidal cells) and of dorsal hippocampal (DHC) dentate granule cells of less anxious (LA) and more (extremely) anxious (MA) stressed animals (PSLA and PSMA). Handled controls (HC) which were less anxious (HCLA) and spontaneously more anxious (HCMA) equivalently to predator stressed subgroups were also studied. Golgi analysis revealed BLA output neurons of HCMA rats exhibited longer, more branched dendrites with higher spine density than the other groups of rats, which did not differ. Finally, spine density of DHC granule cells was equally depressed in HCMA and PSMA rats relative to HCLA and PSLA rats. Total dendritic length of BLA pyramidal and stellate cells (positive predictor) and DHC spine density (negative predictor) together accounted for 96% of the variance of anxiety of handled rats. DHC spine density was a negative predictor of PSMA and PSLA anxiety, accounting for 70% of the variance. Data are discussed in the context of morphological differences as phenotypic markers of a genetic predisposition to anxiety in handled controls, and a possible genetic vulnerability to predator stress expressed as reduced spine density in the DHC. Significance of findings for animal models of anxiety and hyperarousal comorbidities of PTSD are discussed.
Anxiety and Dendritic Spine Density in the Granule Cells of the Rat Hippocampal Dentate Gyrus: Implications for PTSD
Presented at the Society for Neuroscience, Washington, DC (2011)
Noor Shakfeh (1) , Natalie Meylan (2) , Ronald F. Mervis (2, 3) , Robert Adamec (4)
Abstract
Post-Traumatic Stress Disorder (PTSD) is an anxiety response to severe stress. Not all individuals exposed to the same stressor show a long-lasting fear response. Using a rat model, the goal of this study was to ascertain the relationship between innate anxiety levels and a morphological indicator of hippocampal brain circuitry: the spine density on granule cells of the dentate gyrus of the dorsal hippocampus.
Synaptic density may be altered in the presence of various stressors. In a rat, predator stress (e.g, exposure to a cat). is an intense stressor The response to predator stress is also contingent upon whether the subject’s innate level of anxiety is low (e.g, subject is well adapted) or high (e.g., mal-adapted). Anxiety was previously determined by the rat’s response in an elevated plus maze. In the present study, we investigated if less anxious (LA) and more anxious (MA) stressed and unstressed animals differed in the dendritic spine density of granule cells of the dentate gyrus of the dorsal hippocampus from right and left hemispheres.
Two weeks after exposure to predator stress (or non-stress “handled” conditions), the rats were sacrificed. The fixed brain tissue was Golgi stained and coded slides prepared. Dendritic spine density was assessed on randomly selected granule cells of the dorsal hippocampal dentate gyrus from both hemispheres.
Analysis showed that spine density of the granule cells was bilaterally and equally depressed in both high anxious groups (stressed and unstressed) relative to the low anxious groups. Thus, regardless of exposure to a stressor, the granule cells from “high anxious” (= mal-adapted) rats had significantly less spine density (- 10-14%) than from the “less anxious” (= well-adapted) rats.
These results suggest that altered hippocampal circuitry (partially a function of dendritic spine density) in the mal-adapted (high anxiety) rats may represent the basis for a possible genetic vulnerability to stress anxiety, a co-morbid symptom of PTSD.
SIRT1 Is Essential for Normal Cognitive Function and Synaptic Plasticity
- J Neurosci. 2010 Jul 21;30(29):9695-707
- doi: 10.1523/JNEUROSCI.0027-10.2010.
Shaday Michan,1,3* Ying Li,4,5* Maggie Meng-Hsiu Chou,4 Edoardo Parrella,5 Huanying Ge,6 Jeffrey M. Long,7 Joanne S. Allard,7 Kaitlyn Lewis,7 Marshall Miller,7 Wei Xu,4 Ronald F. Mervis,8,9 Jing Chen,2 Karen I. Guerin,2 Lois E. H. Smith,2 Michael W. McBurney,10 David A. Sinclair,1 Michel Baudry,4 Rafael de Cabo,7 and Valter D. Longo4,5
Abstract
Conservation of normal cognitive functions relies on the proper performance of the nervous system at the cellular and molecular level. The mammalian nicotinamide-adenine dinucleotide-dependent deacetylase SIRT1 impacts different processes potentially involved in the maintenance of brain integrity, such as chromatin remodeling, DNA repair, cell survival, and neurogenesis. Here we show that SIRT1 is expressed in neurons of the hippocampus, a key structure in learning and memory. Using a combination of behavioral and electrophysiological paradigms, we analyzed the effects of SIRT1 deficiency and overexpression on mouse learning and memory as well as on synaptic plasticity. We demonstrated that the absence of SIRT1 impaired cognitive abilities, including immediate memory, classical conditioning, and spatial learning. In addition, we found that the cognitive deficits in SIRT1 knock-out (KO) mice were associated with defects in synaptic plasticity without alterations in basal synaptic transmission or NMDA receptor function. Brains of SIRT1-KO mice exhibited normal morphology and dendritic spine structure but displayed a decrease in dendritic branching, branch length, and complexity of neuronal dendritic arbors. Also, a decrease in extracellular signal-regulated kinase 1/2 phosphorylation and altered expression of hippocampal genes involved in synaptic function, lipid metabolism, and myelination were detected in SIRT1-KO mice. In contrast, mice with high levels of SIRT1 expression in brain exhibited regular synaptic plasticity and memory. We conclude that SIRT1 is indispensable for normal learning, memory, and synaptic plasticity in mice.
Dendritic Alterations in the Frontal and Parietal Cortices in Mild Cognitive Impairment and Alzheimer’s disease.
Presented at the American Society for Neural Transplantation and Repair, Clearwater Beach, Florida, (2009)
Ronald F. Mervis1,2,3 , James Kotick3,4, Mrunal Shah3,4, Adam Winkler5, Konstance Kasimos4, Siddharth Joshi4 , Stephen Scheff 6, Elliott J. Mufson7
Abstract
Cortical dendritic morphology (i.e. branching and spine configuration) may play an important role in Alzheimer’s disease (AD)-related cognitive decline. This study assessed dendritic morphology in three clinical groups: Individuals with no cognitive impairment (NCI), mild cognitive impairment (MCI) and mild to moderate AD. Formalin-fixed tissue blocks from the frontal and (area 9) and the parietal cortex (area 39-40) were stained with the Golgi method, and layer II-III pyramidal neurons were randomly selected for analysis by an investigator blinded to clinical status using coded slides. Dendritic analysis in the parietal cortex revealed a significant progressive loss of branching that paralleled increasing cognitive deficits: e.g., NCI > MCI > AD. In frontal cortex, MCI pyramidal neurons manifested increased dendritic branching and spine number compared to NCI with a subsequent significant reduction of these parameters in AD. This transient increase in branching and spines may represent a compensatory neuroplastic response that may assist in maintaining cortical circuitry and delay more severe cognitive dysfunction in MCI. Although the basis for this increase in MCI is unknown, mutations in amyloid beta (Aß) and/or tau genes may enhance dendritic pruning (microtubule depolymerization in the shafts of dendrites) by promoting enhanced calcium influx from the endoplasmic reticulum, or Aß could promote spine formation via actin polymerization.
Supported by AG14449.
Neurotoxic Consequences of Intraneuronal Aβ Oligomeric Aggregates on Hippocampal Dendritic Morphology in the 3 month old ArcAβ Transgenic Mouse
Presented at the Alzheimers Association International Conference, Prague (2009)
*R. F. MERVIS1,2, M. KNOBLOCH3, N. JANI4, S. MORADIAN4, B. A. THOMAS2, L. NATTKEMPER2,5, U. KONIETZKO6, R. M. NITSCH6;
Abstract
The ArcAβ transgenic mouse expresses human APP695 with combined Swedish and Arctic mutations. This results in a transgenic mouse model of brain β-amyloid pathology caused by both increased production of Aβ and enhanced formation of intraneuronal oligomeric Aβ species. These intraneuronal aggregates occur as early as 3 months and precede amyloid plaque formation. The purpose of this study was to compare dendritic branching and spines in the hippocampus of young (3.5 mon-old) ArcAβ mice to age-matched wild type controls and to assess the effects of these early small intraneuronal aggregates on dendritic circuitry. Formalin-fixed brains (5 per group) were stained using the Rapid Golgi method. All slides were coded. Camera lucida drawings of dendritic arbors of randomly selected CA1s and granule cells of the dentate gyrus were quantified using Sholl analysis and dendritic spine density was assessed. Compared to controls, in the young ArcAβ Tg mice there was a reduction in dendritic branching of the hippocampal CA1s (-34%) and in the granule cell arbors (-15%). This was accompanied by a loss of dendritic spines on CA1s (-22%) and granule cells (-15%). All of these changes were statistically significant. These dendritic parameters were affected before the deposition of extracellular β-amyloid plaques but coincided with the development of intraneuronal Aβ oligomeric aggregates. Findings suggest that the oligomers had a neurotoxic impact on the hippocampal neurons and this resulted in disruption and loss of hippocampal circuitry in these young ArcAβ transgenic mice. This would contribute to the subsequent functional impairment found in these animals.
Decreased number of interneurons and increased seizures in neuropilin 2 deficient mice: Implications for autism and epilepsy
- Epilepsia. 2009 Apr; 50(4): 629–645.
- Published online 2008 Jul 24.
- doi: 10.1111/j.1528-1167.2008.01725.x
John C. Gant* , Oliver Thibault* , Eric M. Blalock* , Jun Yang†, Adam Bachstetter‡, James Kotick‡, Paula E. Schauwecker§, Kurt F. Hauser¶, George M. Smith†, Ron Mervis‡, YanFang Li#,**, and Gregory N. Barnes#,**
Summary
Purpose—Clinically, perturbations in the semaphorin signaling system have been associated with autism and epilepsy. The semaphorins have been implicated in guidance, migration, differentiation, and synaptic plasticity of neurons. The semaphorin 3F (Sema3F) ligand and its receptor, neuropilin 2 (NPN2) are highly expressed within limbic areas. NPN2 signaling may intimately direct the apposition of presynaptic and postsynaptic locations, facilitating the development and maturity of hippocampal synaptic function. To further understand the role of NPN2 signaling in central nevous system (CNS) plasticity, structural and functional alterations were assessed in NPN2 deficient mice.
Methods—In NPN2 deficient mice, we measured seizure susceptibility after kainic acid or pentylenetetrazol, neuronal excitability and synaptic throughput in slice preparations, principal and interneuron cell counts with immunocytochemical protocols, synaptosomal protein levels with immunoblots, and dendritic morphology with Golgi-staining.
Results—NPN2 deficient mice had shorter seizure latencies, increased vulnerability to seizurerelated death, were more likely to develop spontaneous recurrent seizure activity after chemical challenge, and had an increased slope on input/output curves. Principal cell counts were unchanged, but GABA, parvalbumin, and neuropeptide Y interneuron cell counts were significantly reduced. Synaptosomal NPN2 protein levels and total number of GABAergic synapses were decreased in agene dose-dependent fashion. CA1 pyramidal cells showed reduced dendritic length and complexity, as well as an increased number of dendritic spines.
Discussion—These data suggest the novel hypothesis that the Sema 3F signaling system’s role in appropriate placement of subsets of hippocampal interneurons has critical downstream consequences for hippocampal function, resulting in a more seizure susceptible phenotype.
Developmental Exposure to Polychlorinated Biphenyls Interferes With Experience-Dependent Dendritic Plasticity and Ryanodine Receptor Expression in Weanling Rats
- Environ Health Perspect. 2009 Mar;117(3):426-35.
- doi: 10.1289/ehp.11771.
1Center for Research on Occupational and Environmental Toxicology, Oregon Health & Science University, Portland, Oregon, USA; 2Veterinary Molecular Biosciences and Center for Children’s Environmental Health, University of California, Davis, California, USA; 3Neurostructural Research Labs and Center for Aging and Brain Repair, University of South Florida College of Medicine, Tampa, Florida, USA; 4Neurotoxicology Division, National Health and Environmental Effects Research Laboratory, U.S. Environmental Protection Agency, Research Triangle Park, North Carolina, USA; 5Department of Psychology, Florida Atlantic University, Boca Raton, Florida, USA; 6Department of Pediatrics, University of Oklahoma Health Sciences Center, Oklahoma City, Oklahoma, USA; 7Department of Molecular Microbiology and Immunology, Johns Hopkins Bloomberg School of Public Health, Baltimore, Maryland, USA; 8Department of Environmental & Molecular Toxicology, Oregon State University, Corvallis, Oregon; USA; 9Department of Veterinary and Comparative Anatomy, Physiology and Pharmacology, College of Veterinary Medicine, Washington State University, Pullman, Washington, USA; 10Department of Environmental Health Science, Johns Hopkins Bloomberg School of Public Health, Baltimore, Maryland, USA
Background: Neurodevelopmental disorders are associated with altered patterns of neuronal connectivity. A critical determinant of neuronal connectivity is the dendritic morphology of individual neurons, which is shaped by experience. The identification of environmental exposures that interfere with dendritic growth and plasticity may, therefore, provide insight into environmental risk factors for neurodevelopmental disorders.
Objective: We tested the hypothesis that polychlorinated biphenyls (PCBs) alter dendritic growth and/or plasticity by promoting the activity of ryanodine receptors (RyRs).
Methods and results: The Morris water maze was used to induce experience-dependent neural plasticity in weanling rats exposed to either vehicle or Aroclor 1254 (A1254) in the maternal diet throughout gestation and lactation. Developmental A1254 exposure promoted dendritic growth in cerebellar Purkinje cells and neocortical pyramidal neurons among untrained animals but attenuated or reversed experience-dependent dendritic growth among maze-trained littermates. These structural changes coincided with subtle deficits in spatial learning and memory, increased [ 3H]-ryanodine binding sites and RyR expression in the cerebellum of untrained animals, and inhibition of training-induced RyR upregulation. A congener with potent RyR activity, PCB95, but not a congener with negligible RyR activity, PCB66, promoted dendritic growth in primary cortical neuron cultures and this effect was blocked by pharmacologic antagonism of RyR activity.
Conclusions: Developmental exposure to PCBs interferes with normal patterns of dendritic growth and plasticity, and these effects may be linked to changes in RyR expression and function. These findings identify PCBs as candidate environmental risk factors for neurodevelopmental disorders, especially in children with heritable deficits in calcium signaling.
Activation of estrogen receptor-β regulates hippocampal synaptic plasticity and improves memory
Abstract
Estrogens have long been implicated in influencing cognitive processes, yet the molecular mechanisms underlying these effects and the roles of the estrogen receptors alpha (ERα) and beta (ERβ) remain unclear. Using pharmacological, biochemical and behavioral techniques, we demonstrate that the effects of estrogen on hippocampal synaptic plasticity and memory are mediated through ERβ. Selective ERβ agonists increased key synaptic proteins in vivo, including PSD-95, synaptophysin and the AMPA-receptor subunit GluR1. These effects were absent in ERβ knockout mice. In hippocampal slices, ERβ activation enhanced long-term potentiation, an effect that was absent in slices from ERβ knockout mice. ERβ activation induced morphological changes in hippocampal neurons in vivo, including increased dendritic branching and increased density of mushroom-type spines. An ERβ agonist, but not an ERα agonist, also improved performance in hippocampus-dependent memory tasks. Our data suggest that activation of ERβ can regulate hippocampal synaptic plasticity and improve hippocampus-dependent cognition.
Persistent Neurostructural Consequences of Prenatal Exposure to Moderate Doses of Cannabinoids in the Rat.
Abstract
Marijuana is the most widely used illegal drug among women of reproductive age, Reports dealing with the effects of prenatal exposure to this substance of abuse are still controversial. In particular, the implications of possible long-term consequences of in utero exposure to cannabis derivatives on brain circuitry are poorly understood.In this study the synthetic CB1 agonist WIN 55,212-2 (WIN) was administered daily to pregnant rats from gestational day 5 to 20 (0.5mg/kg). This dose is equivalent to a moderate or low exposure to marijuana in humans and has no overt toxic effects. The treatment with WIN did not affect gestational and reproduction parameters and WIN-exposed pups did not show any sign of malformations or malnutrition. Behaviorally, however, prenatal treatment with WIN altered pup performance in homing behavior and produced a decrease in the rate of separation-induced ultrasonic vocalizations. Behavioral deficits that resulted were long-lasting, since prenatal WIN exposure caused a disruption of memory retention in young and adult offspring subjected either to a passive or an active avoidance task. Moreover, an analysis of dendritic branching of Golgi-impregnated hippocampal granule cells of the dentate gyrus in prenatal WIN-exposed rats sacrificed at 40 and 80 days-old showed a significant increase in the amount of dendritic arbor and in the complexity of the dendritic trees at both ages compared to age-matched controls.. These findings suggest that moderate exposure to cannabinoids during crucial periods of brain development can cause dysmorphic maturation of the hippocampus associated with the failure of these neurons to undergo normal age-related dendritic pruning. Such subtle morphological alterations and commensurate changes in brain circuitry would be, in turn, a factor underlying the behavioral deficits observed both in early and late postnatal life.
Hyperglycemia Not Hypoglycemia Alters Neuronal Dendrites and Impairs Spatial Memory
Abstract
Background/objective: We previously reported that chronic hyperglycemia, but not hypoglycemia, was associated with the reduction of neuronal size in the rat brain. We hypothesized that hyperglycemia-induced changes in neuronal structure would have negative consequences, such as impaired learning and memory. We therefore assessed the effects of hyperglycemia and hypoglycemia on neuronal dendritic structure and cognitive functioning in young rats.
Design/methods: Experimental manipulations were conducted on male Wistar rats for 8 wk, beginning at 4 wk of age. At the completion of the treatments, all rats were trained in the radial-arm water maze, a spatial (hippocampus-dependent) learning and memory task. Three groups of rats were tested: an untreated control group, a streptozotocin-induced diabetic (STZ-D) group, and an intermittent hypoglycemic group. Following behavioral training, the brains of all animals were examined with histologic and biochemical measurements.
Results: Peripheral hyperglycemia was associated with significant increases in brain sorbitol (7.5 +/- 1.6 vs. 5.84 +/- 1.0 microM/mg) and inositol (9.6 +/- 1.4 vs. 7.1 +/- 1.1 microM/mg) and reduced taurine (0.65 +/- 0.1 vs. 1.3 +/- 0.1 mg/mg). Histologic evaluation revealed neurons with reduced dendritic branching and spine density in STZ-D rats but not in control or hypoglycemic animals. In addition, the STZ-D group exhibited impaired performance on the water maze memory test.
Conclusions: Hyperglycemia, but not hypoglycemia, was associated with adverse effects on the brain polyol pathway activity, neuronal structural changes, and impaired long-term spatial memory. This finding suggests that the hyperglycemic component of diabetes mellitus has a greater adverse effect on brain functioning than does intermittent hypoglycemia.
Chronic Methamphetamine Induces Structural Changes in Frontal Cortex Neurons and Upregulates Type I Interferons
Abstract
While methamphetamine-induced changes in brain neurotransmitters, their receptors and transporters are well studied; the means by which methamphetamine abuse results in cognitive and behavioral abnormalities is unknown. Here we administered methamphetamine chronically, in doses relevant to recreational usage patterns, to nonhuman primates. Neurostructural analysis revealed decreased dendritic material and loss of spines in frontal lobe neurons. Molecular examination demonstrated that type I interferons (interferon-alpha and -beta) increased in the frontal lobe in response to chronic methamphetamine treatment, in correlation with the neuronal changes. Chronic methamphetamine thus results in significant changes in the primate brain, inducing cytokines and altering neuronal structure, both of which can contribute to functional abnormalities.
Effects of a blueberry enriched diet on dendritic parameters of striatal neurons in the aging rat brain: a paradoxical conundrum.
Abstract
Blueberries (BB) contain nutritional antioxidants (e.g., polyphenols) which may attenuate neuronal damage stemming from age-related oxidative stress and the formation of reactive oxygen species. BB also may exert anti-inflammatory and modify cell signaling mechanisms. During aging, cortical pyramidal neurons show a loss of dendritic arbor and of spines. Previously, we found that a BB-enriched diet could increase dendritic branching and spines in the older rats. Here, we evaluated these dendritic parameters in medium spiny neurons in the rat striatum. In rodents, these neurons apparently continue to show expansion of their dendritic domains until late in old age.
This study evaluated the effects of a 2% BB dietary supplement on striatal dendritic parameters in aging rats. Nineteen month old male Fischer 344 rats were given either a standard NIH-31 rat chow (control subjects) or an NIH chow enriched with 2% BB extract for 2.5 months. Following behavioral testing, the 21.5 mon-old rats were killed and their brains stained using the Golgi method (N = 3 subjects/group). Brains from a young (6 month-old) control group provided data for an age comparison. From coded slides, randomly selected striatal neurons in the 21.5 mon- old rats were evaluated for the extent of their dendritic branching and density of dendritic spines. Results: Comparison of the 6mo and the 21.5 mo rats showed that with normal aging, there was an increase in the extent and complexity of the dendritic arbor of the striatal neurons. Two and half months of the BB-enriched diet resulted in a decrease in amount of striatal neuron dendritic arbor in the old rats along with a decrease in branching complexity, that is, a change back toward the morphology found in the young animals. There was a significant loss of dendritic spines in the older rats, and while the BB diet did not increase total spines, it did increase L-type spines (which are associated with learning processes). These results are paradoxical in that the effects of the BB diet on the dendritic branching of the striatum in older rats is to drive the morphology of the neurons back toward that seen in younger subjects and this occurs by reducing of the amount of dendritic arbor and branching complexity. This further suggests that even in old subjects, a modest (2%) BB dietary supplementation for 2.5 months appears to exert a significant impact on neuronal morphology and brain circuitry.
Ontogenetic Alterations in Molecular and Structural Correlates of Dendritic Growth After Developmental Exposure to Polychlorinated Biphenyls
OBJECTIVE: Perinatal exposure to polychlorinated biphenyls (PCBs) is associated with decreased IQ scores, impaired learning and memory, psychomotor difficulties, and attentional deficits in children. It is postulated that these neuropsychological deficits reflect altered patterns of neuronal connectivity. To test this hypothesis, we examined the effects of developmental PCB exposure on dendritic growth.
METHODS: Rat dams were gavaged from gestational day 6 through postnatal day (PND) 21 with vehicle (corn oil) or the commercial PCB mixture Aroclor 1254 (6 mg/kg/day). Dendritic growth and molecular markers were examined in pups during development.
RESULTS: Golgi analyses of CA1 hippocampal pyramidal neurons and cerebellar Purkinge cells indicated that developmental exposure to PCBs caused a pronounced age-related increase in dendritic growth. Thus, even though dendritic lengths were significantly attenuated in PCB-treated animals at PND22, the rate of growth was accelerated at later ages such that by PND60, dendritic growth was comparable to or even exceeded that observed in vehicle controls. Quantitative reverse transcriptase polymerase chain reaction analyses demonstrated that from PND4 through PND21, PCBs generally increased expression of both spinophilin and RC3/neurogranin mRNA in the hippocampus, cerebellum, and cortex with the most significant increases observed in the cortex.
CONCLUSIONS: This study demonstrates that developmental PCB exposure alters the ontogenetic profile of dendritogenesis in critical brain regions, supporting the hypothesis that disruption of neuronal connectivity contributes to neuropsychological deficits seen in exposed children
Enrichment Improves Cognition in AD Mice by Amyloid-Related and Unrelated Mechanisms
Abstract
Lifelong cognitive stimulation is associated with a lower risk of Alzheimer’s disease (AD), but causality is difficult to prove. We therefore sought to investigate the preventative potential of environmental enrichment (EE) using mice expressing both human mutant presenilin-1 and the amyloid precursor protein (PS1/PDAPP). At weaning, mice were placed into either an enriched or standard housing environment. Behavioral testing at 4.5–6 months showed that environmentally enriched PS1/PDAPP mice outperformed mice in standard housing, and were behaviorally indistinguishable from non-transgenic mice across multiple cognitive domains. PS1/PDAPP mice exposed to both environmental enrichment and behavioral testing, but not to EE alone, showed 50% less brain β-amyloid without improved dendritic morphology. Microarray analysis revealed large enrichment-induced changes in hippocampal expression of genes/proteins related to Aβ sequestration and synaptic plasticity. These results indicate that EE protects against cognitive impairment in AD transgenic mice through a dual mechanism, including both amyloid dependent and independent mechanisms.
Influence of predator stress on the consolidation versus retrieval of long-term spatial memory and hippocampal spinogenesis.
Abstract
We have studied the influence of predator stress (30 min of cat exposure) on long-term (24 h) spatial memory and the density of spines in basilar dendrites of CA1 neurons. Predator stress occurred either immediately before water maze training (Stress Pre-Training) or before the 24 h memory test (Stress Pre-Retrieval). The Control (nonstress) group exhibited excellent long-term spatial memory and a robust increase in the density of stubby, but not mushroom, shaped spines. The Stress Pre-Training group had impaired long-term memory and did not exhibit any changes in spine density. The Stress Pre-Retrieval group was also impaired in long-term memory performance, but this group exhibited an increase in the density of stubby, but not mushroom, shaped spines, which was indistinguishable from the control group. These findings indicate that: (1) A single day of water maze training under control conditions produced intact long-term memory and an increase in the density of stubby spines in CA1; (2) Stress before training interfered with the consolidation of information into long-term memory and suppressed the training-induced increase in spine density; and (3) Stress immediately before the 24 h memory test trial impaired the retrieval of the stored memory, but did not reverse the training-induced increase in CA1 spine density. Overall, this work provides evidence of structural plasticity in dendrites of CA1 neurons which may be involved in the consolidation process, and how spinogenesis and memory are modulated by stress.
Exposure to Depleted Uranium Results in CA1 Dendritic Neuroplasticity in the Adult Rat Hippocampus
Abstract
Gulf War I veterans with long-term residual depleted uranium (DU) shrapnel were found to have lowered performance efficiency in selected neurocognitive tests. Studies of rats with intramuscular implantation of depleted uranium (DU) reveal that in addition to accumulation of this metal in kidney and bone, it is also increased in brain. This study was undertaken to assess the neurotoxicologic effects of implanted DU in adult rats, focusing on dendritic changes to the hippocampal pyramidal cells that could affect memory.
Adult male Sprague-Dawley rats each had 20 DU or tantalum (control) pellets (1mm x 2mm) implanted in the gastrocnemius muscles, for a six month period. Upon sacrifice, coronal hemispheric blocks were removed and stained by the Rapid Golgi impregnation procedure. From coded slides, the basilar dendritic arbor of randomly selected hippocampal CA1 pyramidal cells was quantitatively assessed using Sholl analysis. Results show that — relative to controls — there was a significant increase (+23%, p < .0005, Wilcoxon test) in dendritic material in the inner 1/2 of the arbor in rats administered DU. There was no difference between groups for the outer half of the CA1 dendritic arbor.
Thus, in adult rats, 24 weeks exposure to a high dose of implanted DU pellets caused an abnormal dendritic neuroplastic effect on CA1 branching and altered hippocampal circuitry. This may be the neuroanatomical basis for the deleterious cognitive sequelae described for the Gulf War soldiers. (Supported by DAMD17-01-1-0775, US Army Medical Research and Materiel Command)
Cardiac arrest with cardiopulmonary resuscitation reduces dendritic spine density in CA1 pyramidal cells and selectively alters acquisition of spatial memory.
Abstract
The hippocampus is highly sensitive to ischemia and is one of the most extensively damaged regions of brain during cardiac arrest. Damage to hippocampus can subsequently lead to learning and memory deficits. The current study used the Morris water maze to characterize spatial learning and memory deficits elicited by 8 min of cardiac arrest with cardiopulmonary resuscitation (CA/CPR) in mice, which is associated with a 25-50% decrease in CA1 neurons. Mice were trained to navigate the water maze prior to CA/CPR or sham surgery (SHAM). They were retested in the water maze on days 7 and 8 postsurgery; both CA/CPR and SHAM groups were able to perform the task at presurgical levels. However, when the hidden platform was moved to a new location, the SHAM mice were able to adapt more quickly to the change and swam a shorter distance in search of the platform than did CA/CPR mice. Thus, CA/CPR did not affect the ability of mice to retain a previously learned platform location, but it did affect their ability to learn a new platform location. This behavioural impairment was correlated with dendritic spine density in the CA1 region of the hippocampus. Data presented here suggest that morphological changes, such as spine density, that occur in neurons that survive CA/CPR may be associated with cognitive impairments
Insulin-like Growth Factor 1 Is Essential for Normal Dendritic Growth
Abstract
This study evaluated somatic and dendritic growth of neurons in the frontoparietal cortex of Igf1-/- brains. Pyramidal neuron density was increased by approximately 25% (P =.005) and soma size reduced by approximately 10% (P <.001). Golgi staining revealed that cortical layer II-III neurons exhibited a significant reduction in dendritic length and complexity in Igf1 null mice. Dendritic spine density and presumably synaptic contacts were reduced by 16% (P =.002). Similar findings were obtained for cortical layer V and piriform cortex pyramids. Supporting a reduction in synapses, synaptotagmin levels were reduced by 30% (P <.02) in the Igf1 null brain. Investigation of factors critically involved in dendritic growth and synaptogenesis showed an approximately 50% reduction in cortical CDC42 protein expression (P <.001) and an approximately 10% reduction in brain cholesterol levels (P <.01) in Igf1 null mice. Evidence is presented that Igf1 deletion causes disruptions in lipid and microtubule metabolism, leading to impaired neuronal somatic and dendritic growth.
Prolonged intermittent hypoxia leads to disruption of spatial memory task acquisition in the absence of obvious structural abnormalities in hippocampal CA1 pyramidal neurons.
Abstract
Intermittent hypoxia (IH), a hallmark of obstructive sleep apnea, leads to increased apoptosis in CA1 neurons, peaking at 2 days and returning to control levels at 14 days (IH14), suggesting that surviving neurons were either tolerant or developed tolerance to IH. We examined whether the neurocognitive deficits associated with IH14 were also associated with significant structural changes in pyramidal neurons within CA1. Using Golgi-stained CA1 neurons, camera lucida drawings of the basilar tree were prepared. The extent and distribution of dendritic branching was evaluated by Sholl analysis in 6 IH14 (alternating room air and 10% O2 every 90 sec) and 6 control SD rats. From coded slides 30 randomly selected pyramidal neurons were examined in each group. Although IH14 rats showed significantly prolonged latencies and pathlengths in acquisition of a spatial task in the Morris water maze, no differences were present between IH14 and control rats in the amount and distribution of dendritic material in the 2 groups of neurons. We conclude that those CA1 neurons that exhibit long-term survival in the presence of IH also show normal dendritic branching morphology. We postulate that the spatial memory deficits that develop after IH14 may be due to either reduced neuronal cell populations or altered dendritic spines within CA1s and/or related brain circuits. Supported by NIH HL-63912, HL-66358, HL-65270, and AHA-0050442N
DENDRITIC AND SOMA SIZE ALTERATIONS IN HIPPOCAMPUS AND CORTEX OF RATS FOLLOWING NEONATAL BORNA VIRUS INFECTION
Abstract
Altered neuronal morphology and cell losses are reported in postmortem brain of subjects with autism, including small neurons, decreased dendritic complexity in hippocampus, cerebellar Purkinje cell loss, and at least one report of cortical involvement. Neonatal infection of Lewis rats with Bornavirus, a neurotropic RNA virus, results in neuropathologic, behavioral, and neurochemical abnormalities resembling features of autism. Understanding the processes influencing synaptic dysfunction and selective neuronal losses by apoptosis in this animal model may reveal mechanisms important in neurodevelopmental disorders, including autism. Using Golgi-stained tissue, we evaluated changes in neuronal morphology in dentate gyrus and layer V of fronto-parietal cortex in neonatally infected rats. At 3 weeks postinfection, dendritic branching was decreased in dentate gyrus granule cells (Sholl analysis, p=0.0004) and in layer V pyramidal cells (p=0.0006). Distal dendrites were especially affected. The soma of the granule cells (but not of cortical neurons) were significantly smaller (p=0.0043). Other changes included branch atrophy, spine loss, and varicosities. These findings suggest a morphologic substrate underlying processes that disrupt synaptic plasticity or increase vulnerability to apoptosis during brain development, and support the use of this animal model for uncovering fundamental aspects of neurodevelopmental disorder pathogenesis.
Supported by: NIH grants K08 MH01608 and RO1 HD37546
NEUROTROPHIC EFFECTS OF THE GLYCOSAMINOGLYCAN C3 ON DENDRITIC ARBORIZATION AND SPINES IN THE ADULT RAT HIPPOCAMPUS: A QUANTITATIVE GOLGI STUDY.
Glycosaminoglycans (GAGs) may be effective as a therapeutic strategy in the treatment of Alzheimer’s Disease (AD). Previous work from this group using a rat model showed that a single intra-amygdaloid injection of Aβ (25-35) could induce abnormal tau-immunoreactive perikarya in the hippocampus. Furthermore, administration of the GAG C3, an ultra low molecular weight heparin mixture of 4-10 oligosaccharides (MW~2.1) could decrease Aβ(25-35)-induced tau-2 immunoreactivity in this model of AD.
In this study, we evaluated the effects of stereotaxic intra-amygdaloid injection of Aβ(25-35) 5nml/3µl)and of C3-treatment (administered subcutaneously, s.c.) on dendritic morphology of Golgi-impregnated CA1 pyramids of the hippocampus. Subjects were young-adult, male F344 rats.
There were four groups:
- the controls were rats with intra-amygdaloid (i-a) injection of vehicle (trifluoroacetic acid, TFA, 3µl) followed by saline (32 days, s.c., 1ml/kg, b.i.d.)(N=10);
- treatment groups consisted of:
- i-a injection of Ab 5 nmol per 3 µl followed by 32 days of saline (s.c., 1ml/kg, b.i.d.)(N=5);
- i-a injection of A b 5 nmol per 3 µl followed by GAG-treatment (32 days of C3, 2.5 mg/kg s.c., b.i.d.)(N=5); and
- i-a injection of vehicle (TFA, 3µl) followed by the C3 GAG (32 days, 2.5 mg/kg s.c. , i.d.)(N=5).
Formalin-fixed coronal blocks of rat frontoparietal cortex, which included the underlying hippocampus, were stained using the Rapid Golgi method. Slides were sectioned at 120 µm. All brains were coded, and 6 well-impregnated CA1 neurons were randomly chosen from each subject from the hemisphere ipsilateral to the intra-amygdaloid injection of the Aβ(25-35)or the vehicle. For analysis of dendritic branching, camera lucida drawings were made of the basilar tree. The drawings were then quantified by the Sholl analysis (method of concentric circles) which provides a profile of the extent and distribution of the dendritic material. Dendritic spines were quantified on 4-5 randomly selected terminal tip segments from each neuron.
Results. Compared to the controls (TFA vehicle), dendritic branching of the CA1s was not diminished by Aβ however, dendritic spine density was significantly attenuated (p<0.001). GAG (C3) treatment in the Aβ-treated animals had only a minor effect on increasing dendritic branching (p=0.039, Wilcoxon signed-rank test) and had no impact on spines. However, C3 GAG-treatment alone (e.g., in the TFA vehicle-injected controls) had a remarkable neurotrophic impact on both dendritic branching and spines. C3 treatment increased total dendritic length by 36% (p<0.001) while increasing branching complexity (p<0.05). Simultaneously, dendritic spine density on the terminal tips was increased by 17%. Combining the C3-induced neurotrophic changes in both branching and spines, it was determined that 32 days of C3-treatment resulted in an overall 61% increase in total synaptic contacts on the basilar tree of the CA1 pyramids in these young-adult rats (p<0.001).
These results indicate that the GAG C3 appears to have a remarkable neurotrophic impact on the hippocampal circuitry of the brain of the normal young-adult rat. The apparent inability of the GAG treatment to produce the same effect in the Aβ-injected AD model may suggest that a more extended time frame (and/or a different dosage) is required for the damaged neurons to respond to C3 treatment. These and other related studies (including the assessment of the C3 treatment on neurons in aged rats) are in progress. In addition, while it possible that C3 treatment could enhance learning or memory, it is also feasible that the extensive neurotrophic response in the young rat may be deleterious to learning (due to the “noise” induced by the presence of inappropriate synapses). This, too, is the subject of future study.
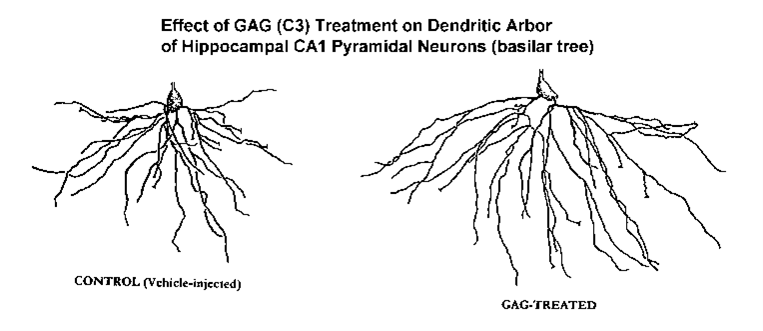
effect on both dendritic branching and spines in the undamaged hippocampus of the adult rat. While awaiting the results of future and more targeted studies, they also suggest that such treatment may be useful in the treatment of AD or in attenuating age-related dendritic atrophy. The study also demonstrates the value of analysis of dendritic branching and spines from Golgi-impregnated tissue to demonstrate and quantify this structural neuroplasticity. (Supported by HHS STTR grant # AG15740-01awarded to RFM and IH)
Hippocampal abnormalities and enhanced excitability in a murine model of human lissencephaly.
Abstract
Human cortical heterotopia and neuronal migration disorders result in epilepsy; however, the precise mechanisms remain elusive. Here we demonstrate severe neuronal dysplasia and heterotopia throughout the granule cell and pyramidal cell layers of mice containing a heterozygous deletion of Lis1, a mouse model of human 17p13.3-linked lissencephaly. Birth-dating analysis using bromodeoxyuridine revealed that neurons in Lis1+/- murine hippocampus are born at the appropriate time but fail in migration to form a defined cell layer. Heterotopic pyramidal neurons in Lis1+/- mice were stunted and possessed fewer dendritic branches, whereas dentate granule cells were hypertrophic and formed spiny basilar dendrites from which the principal axon emerged. Both somatostatin- and parvalbumin-containing inhibitory neurons were heterotopic and displaced into both stratum radiatum and stratum lacunosum-moleculare. Mechanisms of synaptic transmission were severely disrupted, revealing hyperexcitability at Schaffer collateral-CA1 synapses and depression of mossy fiber-CA3 transmission. In addition, the dynamic range of frequency-dependent facilitation of Lis1+/- mossy fiber transmission was less than that of wild type. Consequently, Lis1+/- hippocampi are prone to interictal electrographic seizure activity in an elevated [K(+)](o) model of epilepsy. In Lis1+/- hippocampus, intense interictal bursting was observed on elevation of extracellular potassium to 6.5 mM, a condition that resulted in only minimal bursting in wild type. These anatomical and physiological hippocampal defects may provide a neuronal basis for seizures associated with lissencephaly.
HIPPOCAMPAL DISORGANIZATION AND DENDRITIC ATROPHY FOLLOWING ABNORMAL NEURONAL MIGRATION IN LIS1-DEFICIENT MICE: A QUANTITATIVE ANALYSIS OF GOLGI-STAINED TISSUE.
Abstract
Human Type I lissencephaly is a severe neuronal migration disorder that results from mutation or deletion of the gene encoding the beta subunit of platelet-activating factor acetylhydrolase (Lis1). Mice with a heterozygous deletion of Lis1 show abnormal neuronal migration. In the hippocampus of Lis1+/- mice both normally located, and a “stranded” subgroup of CA1 pyramidal neurons are found. We characterized dendritic parameters of both populations of CA1 neurons. Formalin-fixed coronal blocks from mutants (N=4) and wild type (WT) controls (N=7) were Golgi stained. From coded slides, normally located CA1 pyramids (in the str. pyramidale) and stranded CA1s were randomly selected for analysis. Camera lucida drawings were quantified. Dendritic analysis showed that normally located CA1s in both the KO mice and WT controls were not structurally different. However, stranded CA1s were significantly atrophic and had far more simplistic dendritic arbors. Nevertheless, spine density on these stranded CA1 neurons was normal. Such structural abnormalities may explain in part the reduced electrographic seizure threshold observed in Lis1+/- mice. Supported by NIH.
NEOCORTICAL NEUROPROTECTION BY BONE MORPHOGENIC PROTEIN-7 FOLLOWING MIDDLE CEREBRAL ARTERY OCCLUSION IN THE RAT
Abstract
Bone Morphogenetic Protein-7 (BMP-7) a.k.a Osteogenic protein-1 (OP-1) has previously been shown to enhance recovery of function when administered 24 hours after unilateral middle cerebral artery occlusion (MCAO) in the rat. Using the microscopic appearance of Golgi-stained neuropil as a highly sensitive histopathological technique for assessing tissue damage, we now show that BMP-7 reduced the extent of tissue damage surrounding the infarct.
Adult S-D rats were subjected to permanent right MCAO. Approximately 24 and 72 hours after infarction the animals were administered BMP-7 (~0.4nmol) or vehicle by percutaneous injection into the cisterna magna. Animals were sacrificed 21 days post-MCAO. Brains were immersion-fixed in formalin and 3mm-thick coronal blocks within the infarcted region were removed and stained using the rapid Golgi method.
Neocortex from the infarcted hemisphere was categorized as: (1) missing; (2) heavily damaged (severely necrotic with no Golgi-impregnated neurons); (3) moderate-to-mildly damaged (with well-impregnated neurons and a grainy background in neuropil); and (4) undamaged (impregnated neurons with a clear neuropil). From coded slides, neocortical areas of the different categories were quantified.
BMP-7 treated rats had less tissue missing (p=0.0012) and more lightly-to-moderately damaged (p=0.0098) and undamaged tissue (p=0.001) than control rats. Since Golgi-stained neurons in the moderately-to-lightly damaged penumbral region had less dendritic material (p=0.002) it is possible that this population was characterized by injured cells rescued from subsequent neuronal death by BMP-7. (Funded by Creative BioMolecules, Inc.)
Neurotoxic Effects of Intra-Amygdaloid Injection of A-beta on the Dendritic Arbor of Cingulate Pyramidal Neurons in Adult F344 Rats. A Quantitative Golgi Analysis.
Abstract
A potential animal model of Alzheimer’s disease (AD) was assessed, based on the neurotoxic effects of amyloid-β (25-35) stereotaxically injected directly into the amygdala. Previously, this was found (Sigurdsson et al, 1996) to result in progressive trans-synaptic cytoskeletal and astroglial reactions, including tau2 immunoreactivity distally in cingulate cortex and hippocampus. The present study reveals additional information regarding neurostructural damage of cingulate neurons in this model. Young-adult (3 month-old) F344 rats (N=8) were given a single intra-amygdaloid injection of Aβ (25-35) (5nmol/3ml) and killed 32 days post-operatively. Controls (N=7) were vehicle-injected. Formalin-fixed fronto-parietal tissue blocks were Golgi-stained. Layer V cingulate neurons (6/brain) were randomly selected from coded slides. Camera lucida drawings of the basilar tree were analyzed using Sholl analysis. Results revealed small, but consistent and statistically significant dendritic atrophy in Aβ injected rats (Wilcoxin test,p<.05). The findings lend additional validity to this animal model of AD and suggest that it may be useful in the evaluation of therapeutic strategies. (Supported by NIH/SBIR Grant #1-R41-AG15740-01)
Neuron Specific Patterns of Dendritic Atrophy and Spine Loss in the Hippocampus of Aging PD-APP Transgenic Mice with ABeta Plaques
The PD-APP transgenic (tg) mouse which overexpresses human amyloid precursor protein (APP717VàF) is an animal model of Alzheimer’s Disease (AD). These mice develop A plaques in an age-related manner and have other AD-like and neurodegenerative pathology. Increasing amyloid deposition has not been demonstrated to result in neuronal loss in these mice. However, damaged neurons would initially display dendritic atrophy and other neuritic changes. We characterized dendritic changes in the hippocampus of these aged mice. Coronal blocks of formalin-fixed tissue encompassing the hippocampal formation from 26 month-old PD-APP mice and age-matched non-tg controls were stained using the Rapid Golgi method. From coded slides, CA1 pyramids (n=66) and granule cells (n=67) in the dentate gyrus were randomly selected for dendritic branching analysis. Camera lucida drawings were analyzed using the Sholl Method of Concentric Circles. This produces a profile delineating the extent and distribution of the dendritic field.
- CA1 Pyramids – basilar tree (figure 1)… Branching: The basilar tree of the CA1 pyramids from the PD-APP mice showed significant widespread atrophic changes throughout the entire arbor characterized by both loss of dendritic material and simplification of branching. Spines: there was a 30% loss of total estimated spines on the basilar tree.
- Granule Cells (figure 2)… Branching: The dendritic arbor of the granule cells showed significant atrophic changes specifically restricted only to the outer 2/3rds of the molecular layer. This portion of the dendritic tree receives afferent input from the entorhinal cortex, an area prominently affected in AD. Spines: there was a 45% loss of total estimated spines on the most distal outer 1/3rd of the granule cells in the molecular layer.
Summary: These results suggest that amyloid deposition may contribute to neuronal damage with concomitant disruption of brain circuitry. Our findings are in general agreement with previous studies of hippocampal dendritic atrophy in AD brains. They further lend additional support to the validity of this animal model of AD and justify its preclinical use in testing therapeutic strategies. (This study was supported by Elan Pharmaceuticals)
Dendritic Alterations in Cortical Pyramidal Cells in the Sparse Fur Mouse
Abstract
Ornithine carbamoyltransferase deficiency, an X-linked trait, leads to toxic hyperammonemia in sparse fur (spf/Y) mice. Quantitative analysis of the basilar dendritic tree of layer V pyramidal cells in frontoparietal cortex stained by the Golgi Kopsch method revealed a significant decrease in both the complexity of the dendritic arbor and in dendritic terminal spine density (60%) in spf/Y mice compared with controls. Such reductions may contribute to behavioral dysfunction observed in spf/Y mice.
Analysis of selected Golgi-stained brain regions of tottering, leaner, and compound heterozygous tottering/leaner mice
Presented at The American Association of Veterinary Anatomists, July, 1999
Tottering (tg/tg), leaner (tgla/tgla), and compound heterozygous tottering/leaner (tg/tgla), mutant mice exhibit, to different degrees: ataxia, petit-mal-like epilepsy and an intermittent, myoclonus-like movement disorder. The tottering (tg) and leaner (tgla) mutations occur in the alpha1A calcium channel subunit which is highly expressed by many CNS neurons. This suggests a correlation between calcium channel dysfunction and occurrence of the neurological disorders exhibited by these mice, but the pathogenesis is far from clear. We initiated a morphologic study of the CNS to gain information concerning the influence these mutations have on CNS neurons. We have carried out a quantitative morphometric analysis of tg/tgla cerebellar Purkinje cells, as 90+ % of calcium channels expressed by these cells contain alpha1A subunits. We also examined the cerebral cortex to study neurons in another brain region. Brains from adult mutant and control mice were prepared using the Golgi-Cox method. Decreased branching area and spine density were observed for tg/tgla cerebellar Purkinje cells. However, tg/tgla cerebral layer II/III pyramidal neurons showed significantly larger dendritic arbors compared to controls, but no significant difference in spine density was observed. Cerebral pyramidal neurons may undergo compensatory neuroplastic changes, possibly in response to impaired cerebellar circuitry. Supported by a NIH grant (K08NS01681) to L.C.A.
Assessment of Cortical Dendritic Alterations Following Traumatic Brain Injury in the Rat: A Golgi Study
Traumatic brain injury (TBI) was produced by a controlled pneumatic impactor striking the entire right sensorimotor cortex of the anesthetized rat. Neurologic testing reveals initial deficits with a biphasic pattern of functional behavioral recovery. Golgi-impregnation staining – which permits detailed microscopic assessment of degenerative and regenerative dendritic changes – was used to quantitatively evaluate the basilar trees in superficial (layers II-IV) and deep (layers V-VI) pyramidal cell populations in the impact zone (“damaged region”) and adjacent cortical (“undamaged”) region. Animals were sacrificed at 6 days (n=3) , 3 months (n=3), and 4.5 months (n=1) after the TBI. There were 5 sham controls. A total of 219 neurons were evaluated. Slides were coded and assessments made blind with respect to the post-trauma times. Parameters quantified included Sholl analyses, measurement of total dendritic lengths, and changes in branch segments. The most salient findings were: Six days following TBI, neurons in both damaged and undamaged cortex revealed widespread dendritic atrophy. Deep pyramidal neurons appeared to be more affected than superficial pyramids. Indices of damage included loss of terminal branches, reduction in dendritic length, and significant reductions in Sholl dendritic arbor profiles. At 3 months post-injury, most dendritic parameters still were suppressed in both damaged and undamaged regions. However, there was an increase in terminal tip segments, possibly predicting new neuronal growth. By 4.5 months we find preliminary but compelling evidence of neuroplasticity in both damaged and undamaged cortex, the undamaged region showing the more robust effect. Golgi-staining thus appears to be a valuable tool to help ascertain the extent of neuronal damage and dendritic plasticity and could be useful in evaluating the efficacy of neuroprotective agents . (Supported by U.S. Army contract DAMD-17-93-C-
Enhanced hemicholinium binding and attenuated dendrite branching in cognitively impaired acetylcholinesterase-transgenic mice.
Abstract
In a search for behavioral, neuroanatomical, and metabolic characteristics of Alzheimer’s disease that may result from cholinergic malfunction, we used transgenic mice overexpressing acetylcholinesterase (AChE) mRNA and active enzyme in brain neurons. Mapping by in situ hybridization revealed that transgenic and host AChE mRNAs were distributed similarly. In a Morris water maze working memory paradigm, adult transgenic mice did not display the characteristic improvement found in control mice either between or within test days and spent less time than control mice in the platform zone. In 5-week-old transgenic mice, the basilar dendritic trees of layer 5 pyramidal neurons from the frontoparietal cortex were essentially as developed as in age-matched controls. However, branching totally ceased after this age, whereas in control adults it continued up to at least 7 months. Therefore, dendritic arbors became smaller in adult transgenic mice than those of controls. Furthermore, the average number of spines was significantly lower on dendritic branches of 7-month-old but not 5-week-old transgenics as compared with controls. Binding of tritiated hemicholinium-3, a blocker of the high-affinity choline uptake characteristic of active cholinergic terminals, was over twofold enhanced in the brain of transgenic mice. In contrast, no differences were observed in the mRNA and ligand binding levels of several different subtypes of nicotinic and muscarinic acetylcholine receptors. These findings suggest that three different hallmarks associated with Alzheimer’s disease–namely, progressive cognitive failure, cessation of dendrite branching and spine formation, and enhanced high-affinity choline uptake–are outcomes of cholinergic malfunction.
Nimodipine Facilitates Retention of the Classically Conditioned Nictitating Membrane Response in Aged Rabbits Over Long Retention Intervals
Abstract
Aged rabbits initially underwent 18 days of acquisition of the classically conditioned nictitating membrane response (NMR) using a tone conditioned stimulus (CS) and an air puff unconditioned stimulus (UCS). They were then treated with a low or high dose of nimodipine or a vehicle for 90 days. During this time no further CS-UCS pairings were presented. They underwent testing for retention of the conditioned response (CR) at 30 and 90 days. Retention testing consisted of 20 presentations of the CS alone. Rabbits in the control condition retained 46.4% of their predrug levels of conditioned responding and rabbits receiving the low dose of nimodipine retained 37.3% of their predrug levels after 30 days. After 90 days, retention in these animals declined to 8.1076 and 14.1070, respectively. In contrast, rabbits receiving the high dose of nimodipine retained 85% of their predrug learning at 30 days with little decline at 90 days (77.1%). Nonassociative factors such as sensitivity to the CS or UCS could not explain these effects.
GP120 as an Etiologic Agent for NeuroAIDS: Neurotoxicity and Model Systems
Abstract
The search for an agent that can mediate the symptoms of NeuroAIDS has been directed at gp120, the major envelope protein from HIV. The toxicity associated with gp120 was examined as a model and predictor of the neuropathological and neuropsychiatric manifestations of AIDS. Studies of the neurotoxic effects of purified gp120 on neurons from the rodent CNS cell cultures indicated the following: potent and selective killing of subpopulations of hippocampal neurons; varying potency of gp120s obtained from various HIV isolates; complete and potent protection from gp120 killing action after treatment with peptides related to vasoactive intestinal peptide; and obligatory presence of glia for gp120-related toxicity. Investigations of gp120 treatment of rodents revealed: cortical neurodystrophy with reduced arborizations and swollen processes; delays in developmental behaviors involving motor skills; peptide T prevention or attenuation of the morphological and behavioral deficits/delays produced by administration of gp120; and impairment of learning in the Morris swim maze. In addition, studies of subcutaneously administered, radiolabeled gp120 in neonatal animals demonstrated the presence of toxic fragments of gp120 in the developing brain. With the use of model test systems of non-human derived cell cultures and neonatal rats, we have captured and predicted a number of the morphological and behavioral deficits associated with AIDS. These multi-disciplinary studies of the actions of gp120 and associated fragments in rodents and rodent cells predict that the loss of cognitive and neurological function in patients with AIDS are attributed in part to interference of critical brain functions by the envelope protein, gp120.
HIV Envelope Protein-Induced Neuronal Damage and Retardation of Behavioral Development in Rat Neonates
Abstract
Cognitive and motor impairment are common symptoms among patients infected with the human immunodeficiency virus (HIV), including children who suffer neurological deficits and are frequently developmentally impaired. The HIV envelope protein, gp120, which has been shown to be toxic to neurons in culture, is shed in abundance by infected cells, and thus may play a significant role in the neuropathology of AIDS. To test this possible mechanism, neonatal rats were injected systemically with purified gp120 and the following consequences were observed: (1) radiolabeled gp120 and toxic fragments thereof were recovered in brain homogenates; (2) dystrophic changes were produced in pyramidal neurons of cerebral cortex; (3) retardation was evident in developmental milestones associated with complex motor behaviors. In parallel studies, co-treatment with peptide T, a gp120-derived peptide having a pentapeptide sequence homologous with vasoactive intestinal peptide, prevented or attenuated the morphological damage and behavioral delays associated with gp120 treatment. These studies suggest that gp120 and gp120-derived toxic fragments may contribute to the neurological and neuropsychiatric impairment related to HIV infection, and that peptide T appears to be effective in preventing gp120-associated neurotoxicity in developing rodents.
Exogenous Nerve Growth Factor Reverses Age-Related Structural Changes in Neocortical Neurons in the Aging Rat. A Quantitative Golgi Study
Abstract
The role of chronic exogenous intracerebroventricular administration of nerve growth factor (NGF) on the morphology of layer V pyramidal cell dendrites in aging rats was quantified using Golgi impregnations. Both dendritic branching and dendritic spines from the basilar tree of randomly selected pyramidal neurons of the frontal cortex were evaluated in young control (4-month-old) Fischer 344 rats, in old controls (24-month-old), and in 24-month-old rats administered NGF for 4 weeks. Sholl analysis of basilar dendritic trees showed that neuronal branching in older rats was significantly greater than that in young rats (probably due to compensatory dendritic hypertrophy). The extent of dendritic material in aged rats receiving NGF, however, was identical to that in young rats, that is, the dendritic tree had regressed in size. Dendritic spine response to NGF treatment depended on the region of the dendritic tree sampled. Normal aging resulted in spine loss. However, NGF treatment restored dendritic spine densities to those seen in young controls on terminal tip segments (“plastic” regions). Internal branch segments (“nonplastic” regions) showed no response to NGF. As dendritic spines are thought to represent the neuroanatomic basis of learning and memory, results suggest that NGF can influence the morphology of cortical neurons (probably indirectly via the basal forebrain projections) and therefore may play an efficacious role in the treatment of geriatric cognitive dysfunction and even perhaps in Alzheimer’s disease.
Dietary Restriction Suppresses Age-Related Changes in Dendritic Spines
Abstract
The effects of dietary restriction by every-other-day (EOD) feeding on dendritic spines in the aging rat neocortex were evaluated in Golgi preparations. After weaning, male Wistar rats were offered a 24% protein diet either ad lib (AL) or EOD. AL-fed groups were sacrificed at 6 and 24–25 months of age. EOD-fed groups were sacrificed at 6, 24, and 30 months. To assess the effects of EOD feeding late in life, another group was fed AL for 19 months, then EOD for 5 months and sacrificed when 24 months old. Spine density and configuration were quantified along 20 μm terminal tip segments from the basilar tree of layer V pyramidal cells of the parietal cortex. Evaluation of spine densities from the 6 and 24 months AL-fed groups showed that there was a significant loss of spines with normal aging (−38%). In EOD-fed rats, spine density did not differ significantly from AL age-matched controls at either 6 or 24 months of age. However, spine densities in 24-month-old rats diet restricted late in life and EOD-fed 30-month-old rats were the same as 6-month-old AL-fed controls and EOD 6-month-old rats, an observation suggesting protection of dendritic spines from age-related loss. Spines were categorized as either L-type (lollipop-shaped), which are more prevalent in young adults, or N-type (nubbin). With normal aging (comparing 6- and 24-month-old AL-fed groups) there was a significant decrease in L-type spines. However, all dietarily restricted groups showed retention of L-type spines. The results indicate that age differences in neuronal morphology may be attenuated by chronic dietary restriction initiated after weaning or late in life.
Chronic dietary choline modulates synaptic plasticity in the cerebellar glomeruli of aging mice.
Mech Ageing Dev. 1985 Apr;30(1):1-9. doi: 10.1016/0047-6374(85)90054-5.PMID: 3999812
Abstract
A morphometric investigation was carried out on ethanolic phosphotungstic acid (E-PTA) stained synaptic junctions in the cerebellar glomeruli of adult, old, old choline-deficient and old choline-supplemented mice. Numerical (Nv) and surface (Sv) density as well as average length (L) of the synapses were calculated on 100 pictures per group. A significant reduction of Nv and Sv, as well as an increase of L was found during aging. Choline deficient animals did not show any change as compared to old animals of the same age. In choline supplemented mice Nv and Sv were significantly increased and L significantly decreased, respectively, as compared to old control littermates. No difference was found between adult and choline supplemented mice. In the cerebellar glomeruli only a small fraction of fibers are cholinergic, therefore the present findings support the idea that dietary choline can influence systems other than cholinergic. The possible role of choline supplementation in the modulation of synaptic plasticity via the synthesis and/or turnover of neuronal membrane choline phospholipids, is discussed.
Senescent Microstructural Changes in Rat Cerebellum
Abstract
Senescent changes in Sprague-Dawley rat cerebellar microstructure have been quantified, focusing on the dominant element of cerebellar information processing, the Purkinje cell. In Golgi-Kopsch sections, many 26-month-old Purkinje cells appear defoliated, with small distal dendrites and spiny branchlets being most affected. The mean Purkinje cell area (soma plus dendrites) in computer-oriented sagittal sections is significantly decreased from 20,675 +/- 1,355 micron2/cell in 6-month-old rats to 17,088 +/- 1,107 micron2/cell in 26-month rats. These morphologic changes may be the hallmark of dying cells: in hematoxylin and eosin (H + E)-stained sections from the same rats we also observe a significant senescent decrease in Purkinje neuron density in every vermis lobule examined (lobules II-VII). Overall, the mean number of Purkinje cells/mm of Purkinje cell layer (measured in 10 micron thick sagittal sections) declines from 16.6 +/- 0.8 cells/mm in young rats to 12.5 +/- 0.2 cells/mm in old rats. As Purkinje cells are lost, so too are ethanolic phosphotungstic acid-stained (EPTA) synapses in the upper molecular layer of the cerebellar vermis (lobules VIII-X), and there is a highly significant within-subjects correlation between Purkinje cell density and synaptic density. Overall, synaptic density (in sagittal, 842 micron2 thin sections) decreases significantly from an average of 150,485 +/- 3,641 synapses/mm2 in 6-month rats to an average of 125,000 +/- 4,849 synapses/mm2 in 26-month rats. These changes are consistent with previous electrophysiologic and biochemical data showing age pathology of the cerebellum.
